《Precision Medication》(ISSN 2950-5232)由中国全科医学杂志社与科爱(KeAi)公司于2024年合作推出,是一本开放获取、同行评议的全英文学术期刊,重点关注并快速报道精准用药相关的高水平研究成果,为学术机构药师、医生、科研人员和药企研发人员搭建国际交流平台,推动精准用药学科体系的构建及其与多学科间的融合发展。
Articles
(点击文题阅读原文)
Review
Sagar Trivedi, Manisha Kawadkar, Diksha Pawar, Rishabh Agade, Ujban Husain
Leonard Whye Kit Lim
Original Research
Jinna Yang, Kaimin Guo, Xiaxia Ren, Xiaolian Zhang, Shuang Zhao, Jiansong Fang, Yu Wei, Pengcheng Yang, Wenjia Wang, Hui Wang, Yunhui Hu
Shuai Liu, Shizhao Yuan, Yan Liu, Shuoxian Jia, Yan Zhao, Ziyi Wang, Chunhua Zhou, Jing Yu
Shubin Feng, Juan Du, Xinru Xie, Yiru Li, Chuyuan Yu, Liling Cui, Mingcan Xue, Chenxi Zhang
01 复发性胶质母细胞瘤个体化诊疗的进展与挑战
ABSTRACT:
Recurrent glioblastoma (rGBM) remains one of the most formidable challenges in neuro-oncology due to its aggressive evolution, treatment resistance, and profound intratumoral heterogeneity. Despite advances in multimodal first-line therapy, recurrence is nearly universal and represents a genetically divergent, therapy-adapted malignancy. This review dissects the evolutionary dynamics of rGBM, including clonal selection, therapy-induced mutagenesis, and proneural-to-mesenchymal shifts. It explores the translational potential of longitudinal sampling, circulating tumor DNA, and multi-omics profiling to dynamically monitor tumor progression and resistance emergence. Personalized therapeutic strategies are critically evaluated, including targeted inhibition of EGFR, PI3K/AKT/mTOR, and PDGFR pathways, immunotherapeutic approaches such as CAR T-cell therapy and neoantigen vaccines, and functional drug screening using patient-derived organoids. Moreover, the manuscript highlights innovations in AI-assisted therapy mapping, precision-guided re-irradiation, and adaptive trial designs that redefine individualized care in rGBM. Persistent challenges such as blood-brain barrier penetration, immune evasion, and lack of real-world clinical integration are also addressed. The convergence of high-throughput molecular diagnostics, AI analytics, and targeted therapies underscores a shift from static to dynamic, biomarker-guided interventions. Realizing the full promise of personalized medicine in rGBM demands systemic reforms, multi-disciplinary integration, and equitable clinical adoption.
向下滑动查看更多内容
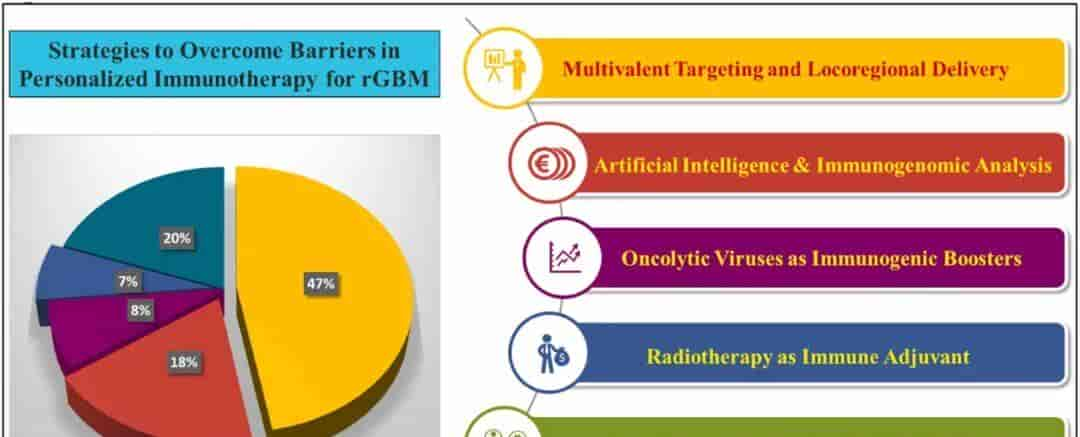
Fig. 1.Strategies to Overcome Barriers in Personalized Immunotherapy for rGBM.
引用文本:
Trivedi S, Kawadkar M, Pawar D, et al. Advances and challenges in personalized diagnosis and therapies for the management of recurrent glioblastoma. Precis Medicat. 2025 Sep;2(3):100052. doi:10.1016/j.prmedi.2025.100052
02
关于新兴人工智能(AI)工具与算法在通过精神益生菌治疗微生物组-肠-脑轴相关疾病中作用的微观综述
ABSTRACT: The microbiome-gut-brain axis involves the bidirectional connection between the gastrointestinal tract, gut microbiome, and central nervous system, and it is pivotal for mental well-being. Imbalances in the gut microbiome (dysbiosis) can disrupt neurotransmitter synthesis, leading to neurodegenerative diseases. Recent researches highlight psychobiotics as effective treatments for mental health conditions like depression. With advancements in artificial intelligence (AI) and large datasets, psychobiotic research has accelerated. AI tools are increasingly used for the prediction, identification, and diagnosis of gut microbiome compositions, enabling faster and more accurate discovery of therapeutic candidates. This review explores the role of AI in enhancing microbiome-gut-brain-axis-related disease therapies through psychobiotics and discusses future directions for this rapidly evolving research field.
向下滑动查看更多内容
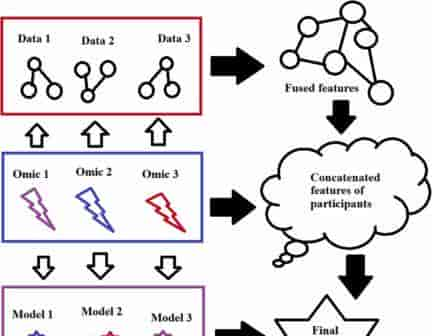
Fig. 1.The graphical workflow of how machine learning model is designed based on omic data.
引用文本:
Lim L W K. A micro review on the role of recently emerged Artificial Intelligence (AI) tools and algorithms in microbiome-gut-brain-axis associated disease therapy via psychobiotics. Precis Medicat. 2025 Sep;2(3):100039. doi:10.1016/j.prmedi.2025.100039
03 阿尔茨海默病药物再定位的多组学整合方法
ABSTRACT: Background: Alzheimer's disease (AD) is a progressive neurodegenerative disorder with an insidious onset, and effective therapeutic agents are urgently needed.
Objective: This study employed a multi-omics integration strategy for drug repurposing against AD.
Methods: Firstly, transcriptomic and proteomic data from AD patients were utilized to identify differentially expressed genes. Potential anti-AD small-molecule compounds were screened by integrating the Reverse Gene Expression Score (RGES) and Connectivity Map (C-Map) approaches with drug-perturbed gene expression profiles from the Library of Integrated Network-Based Cellular Signatures (LINCS), followed by blood-brain barrier (BBB) permeability prediction and structural similarity analysis. Secondly, a drug-disease network was constructed, and Gene Ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway enrichment analyses were performed. The therapeutic potential of candidate drugs was further evaluated via network proximity analysis. Finally, in vitro validation was conducted using Okadaic acid (OA) induced SH-SY5Y and Lipopolysaccharide (LPS) induced BV2 cell models to assess cell viability and nitric oxide (NO) levels. This integrated approach provides a novel framework for identifying repurposed drugs with potential efficacy against AD.
Results: Following the collection of omics data, 227 overlapping candidate compounds were identified through two computational approaches. After BBB prediction screening, 104 drugs were selected for subsequent structural similarity analysis and literature/patent review, ultimately leading to the selection of TNP-470 and Terreic acid for validation. Network pharmacology analysis revealed that potential targets of TNP-470 for AD treatment were significantly enriched in neuroactive ligand-receptor interaction, TNF signaling, and AD-related pathways, while anti-AD targets of Terreic acid primarily involved calcium signaling, AD pathway, and cAMP signaling. Network proximity analysis demonstrated significant associations between both candidates and AD. In vitro assays demonstrated that TNP-470 significantly enhanced the viability of OA-induced SH-SY5Y cells at concentrations of 10 μM and 50 μM (p p .
Conclusion: This drug repositioning strategy based on multi-omics integration provides a novel approach for AD therapeutic development, with both TNP-470 and Terreic acid demonstrating anti-AD potential.
向下滑动查看更多内容
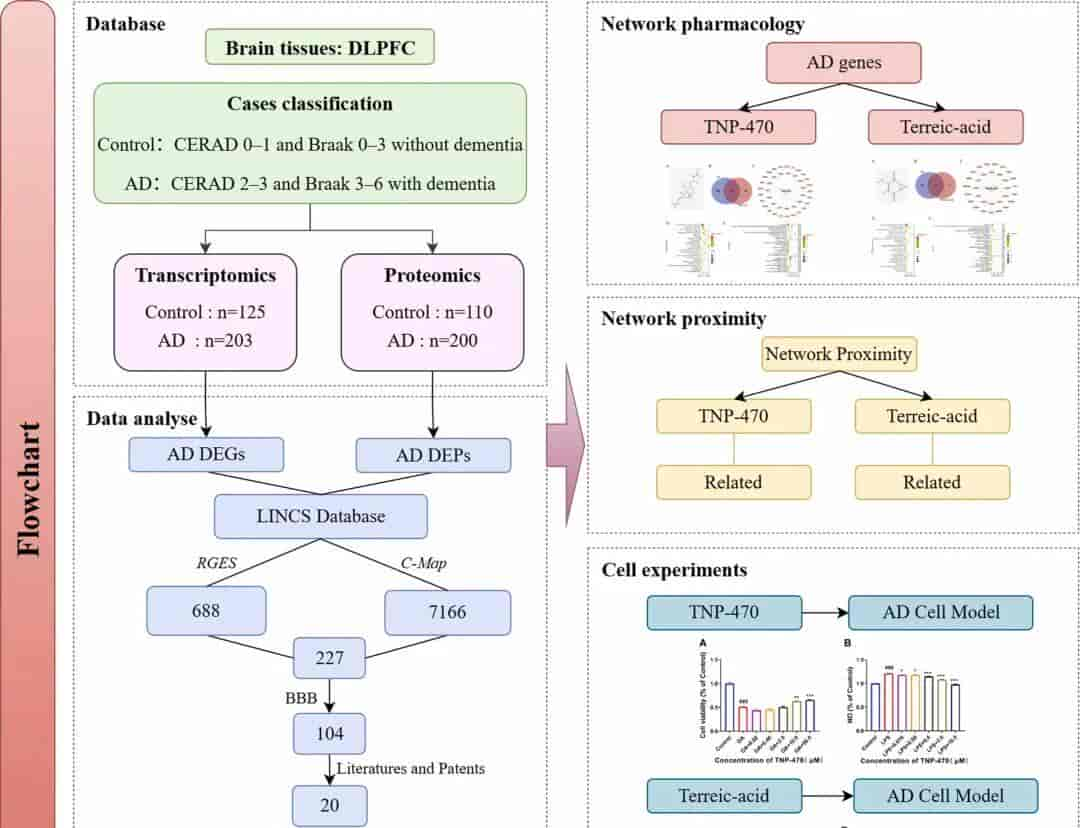
Fig. 1.Flowchart of this study.
引用文本:
Yang J, Guo K, Ren X, et al. Integrative multi-omics approach for drug repositioning in Alzheimer's disease. Precis Medicat. 2025 Sep;2(3):100050. doi:10.1016/j.prmedi.2025.100050
04
不同两性霉素B制剂在侵袭性真菌感染中的有效性、安全性与经济性评价:一项回顾性队列研究

ABSTRACT: Background: Fungal infections have emerged as an increasingly serious public health challenge globally. Four Formulations of amphotericin B are widely used in antifungal therapy. Despite the same active ingredient, they probably differ in efficacy, safety and economics.
Aim: This study aimed to explore the differences in efficacy, safety and economy among different formulations of amphotericin B in patients with IFD.
Methods: We conducted a retrospective study at a tertiary hospital, examining patients who were administered amphotericin B from June 2023 to March 2025, to assess the efficacy, safety and economy of different amphotericin B Formulations in invasive fungal disease.
Results: (1) A total of 71 patients were included. Patients with potential renal injury are more likely to choose liposomal amphotericin B (p = 0.021). (2) Liposomal amphotericin has the accelerated therapeutic onset (p = 0.042), amphotericin B deoxycholate has the delayed therapeutic effect (p = 0.031), the effective response of liposomal amphotericin B in elders was significantly lower (p = 0.022), and the counterpart of amphotericin B deoxycholate in females was significantly higher (p = 0.01). (3) The main adverse events of the three amphotericin B formulations were kidney injury (p .
Conclusion: Amphotericin B formulations exhibit marked variations in efficacy and economy profiles, necessitating individualized selection guided by specific clinical characteristics. Rigorous monitoring of renal function remains imperative throughout the therapeutic course.
向下滑动查看更多内容
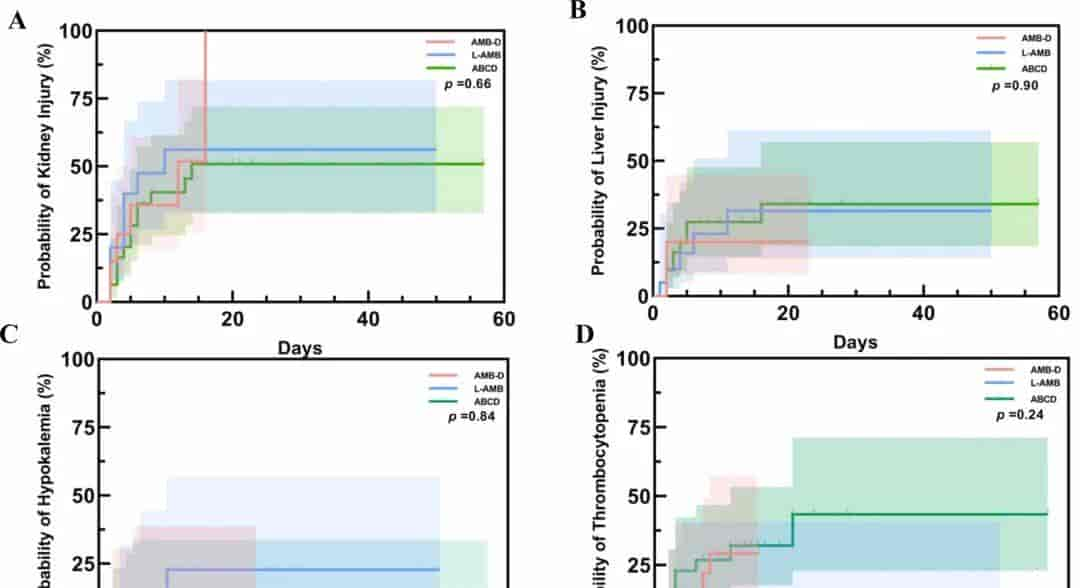
Fig. 4. KM analysis of drug adverse events among different amphotericin B.
引用文本:
Liu S, Yuan S, Liu Y, et al. Evaluation of the efficacy, safety and economy of different amphotericin B formulations in invasive fungal disease: A retrospective cohort study. Precis Medicat. 2025 Sep;2(3):100048. doi:10.1016/j.prmedi.2025.100048
05
近20年神经系统疾病与他汀类药物机制及关联研究的文献计量学分析
ABSTRACT: Background: Neurological disorders are one of the major contributors to the burden of disease, affecting the quality of life of the global population.Statins, known for their cholesterol-lowering properties, have garnered substantial interest due to their potential neuroprotective effects and association with subjective cognitive decline.The study aims to provide a comprehensive bibliometric analysis of the literature on the nexus between neurological disorders and statins.
Methods: Using VOSviewer (1.6.20), CiteSpace (6.3.1 and 5.7.R2), and R (4.4.1), the systematic bibliometric analysis offers valuable insights into the research landscape of neurological disorders and statins from 2004 to 2024.
Results: The findings reveal a notable increase in scholarly output on neurological disorders and statins over the past two decades, with a particular emphasis on clinical trials and pharmacological mechanisms of statins. The remarkable multidisciplinary nature of the field is an important reason for its continued development.Compared to initial exploratory research, recent studies have placed increased emphasis on the neuroprotective roles of statins and their therapeutic potential in managing neurological disorders.Future research is anticipated to focus on precision medicine and personalized therapeutic strategies involving statins.The United States, China and Germany are leading the way in this field of research.
Conclusion: The study's findings are instrumental in informing future research directions and contribute to a broader understanding of the subject matter across various scientific disciplines. Statins have great potential in the treatment of neurological disorders and neuroprotection and are moving towards customized medicine and personalized therapy.
向下滑动查看更多内容
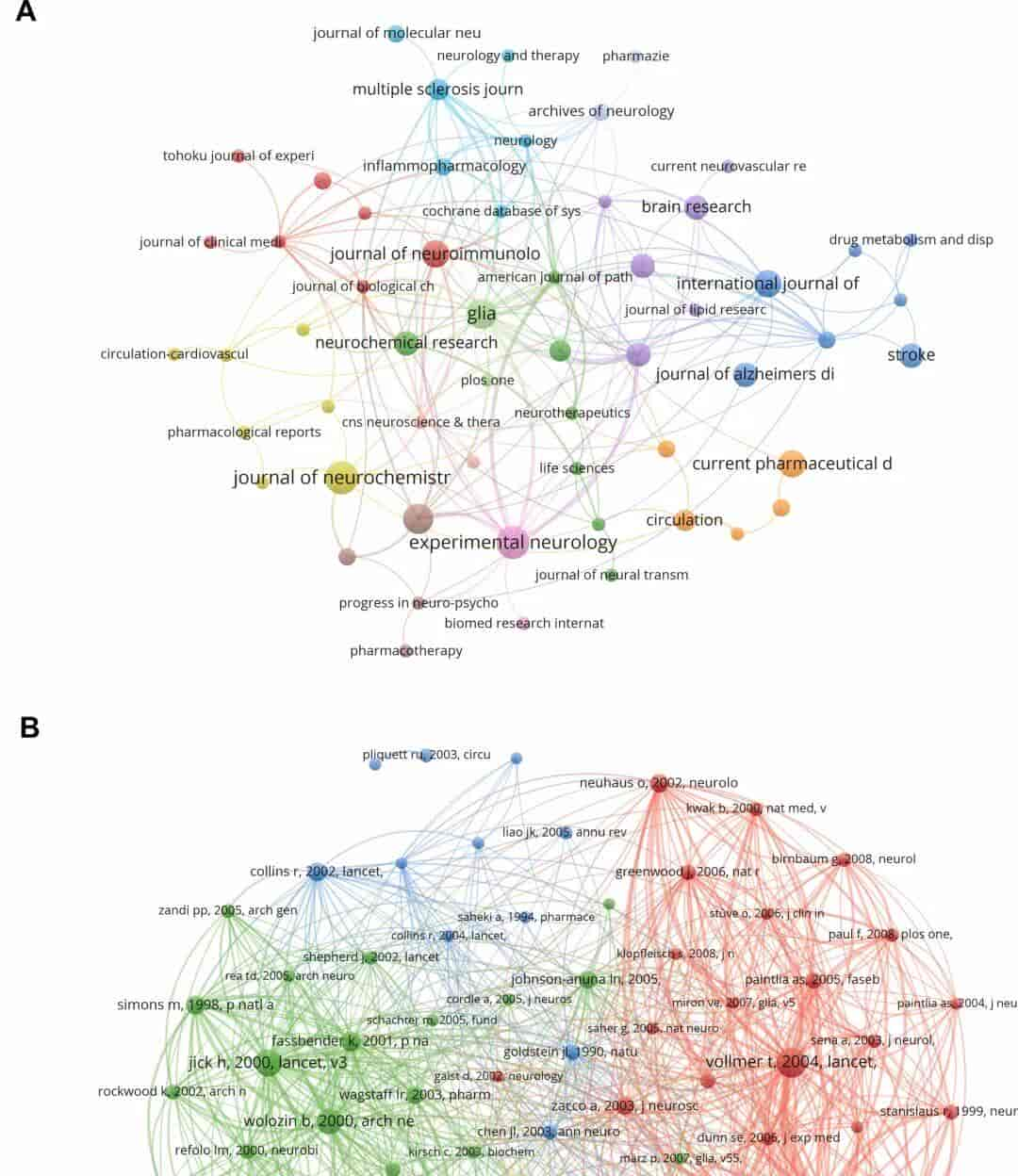
Fig. 4. Research linkages and citation mapping between journals.
引用文本:
Feng S, Du J, Xie X, et al. A bibliometric analysis of mechanisms and linkage studies on neurological disorders and statins over the past 20 years. Precis Medicat. 2025Sep;2(3):100049. doi:10.1016/j.prmedi.2025.100049
文献精读往期回顾
《Precision Medication》征文通知
尊敬的药师、医生以及研究人员:
作为开放获取的同行评议期刊,《Precision Medication》杂志为学术机构药师、医生、科研人员和药企研发人员提供开放、共享的学习交流平台,推动精准用药学科体系的构建及其与多学科间的融合。《Precision Medication》杂志重点关注并快速报道精准用药相关的高水平研究成果,并希冀研究成果在解决临床问题和改善患者结局中应用。我们诚挚邀请您投稿,分享您在精准用药领域的研究成果和见解,以促进该领域的发展。
现面向全球征集学术论文
征稿方向(包括但不限于):
1.精准用药相关政策和指南的发展
2.精准用药研究新方法
3.精准用药体系建设及效果评价
4.数字健康与精准用药
5.信息技术与精准用药
6.药物经济学与精准用药
7.方法学研究
8.药物基因组学研究与临床
9.药物代谢组学研究与临床
10.肿瘤靶向用药
11.新药发现与转化研究
12.慢病精准用药
13.中药精准用药
稿件要求:
1.稿件欢迎双语投稿;也可中文投稿经同行评议被杂志接收后再提供英文稿。
2.优先国家级/省部级基金或其他重大科研基金项目研究、相关临床学科指南/共识及应用研究、大数据及评述等相关论文。
3.原创研究全文不少于5 000字(词)。
投稿方式:
请通过Precision Medication编辑部邮箱提交稿件:pmeditor@precision-med.com.cn
请在投稿时注明“精准用药”。
投稿格式及详细要求请参考Precision Medication官网投稿指南:
我们期待您的宝贵稿件,并期望与全球学术界共同推动精准用药领域的发展!
《Precision Medication》编辑部
《Precision Medication》杂志简介
相关文章